 News
News
-
29
Jan
赵延川课题组核磁共振联盟线上讲座—识别赋能色谱核磁
对真实复杂样品的快速准确检测在食品安全监测、疾病诊断及生物机制研究等方面都有重要的应用。目前色谱分析技术在复杂体系检测中应用最广,但其基于分离,相对耗时,难以满足高通量及原位检测需求。利用光谱及化学传感方法可以避免分离,实现原位快速分析,但分析准确性易受杂质的干扰并且难以获取样品精确的组分信息。色谱方法的“高精确性”和光谱/化学传感方法的“原位及快速性”是分析领域一直追寻的优良检测特性,然而却难以整合在同一种分析技术中。本报告将介绍一种基于核磁共振氟谱的化学传感方法,检测中无需分离,无需标记,实时在线地得到类似于色谱峰的检测信号,精确对应复杂体系中的各个组分。该方法独特的检测能力为研究复杂系统多组分相互作用及复杂化学体系演化规律提供了有力工具。报告也将系统探讨该技术在高通量手性分子检测,生物代谢过程原位跟踪,违禁化学品筛查,动态非共价相互作用量化表征等方面的应用及发展前景。
-
24
Feb
Calix[4]trap: A Bioinspired Host Equipped with Dual Selection Mechanisms
Regulation of recognition events evolving in time and space is vital for living organisms. During ev...
-
28
Jan
Tailoring Sensors and Solvents for Optimal Analysis of Complex Mixtures Via Discriminative 19F NMR Chemosensing
Separation-free analytic techniques capable of providing precise and real-time component information...
 Research Highlight
Research Highlight
-
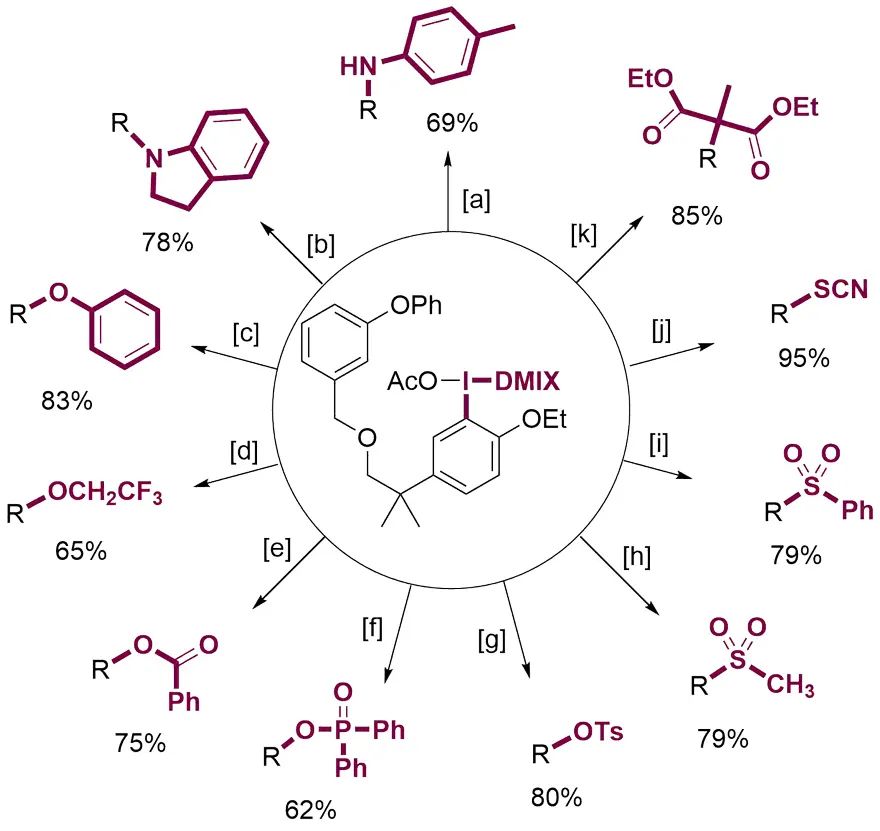
Metal-Free C-H Functionalization via Diaryliodonium Salts with a Chemically Robust Dummy Ligand
A two-step strategy for the transition-metal-free C−H functionalization of arenes using unsymmetrical iodonium salts as versatile synthetic linchpins is presented. The key to the success of this strategy is the identification of the 3,5-dimethyl-4-isoxazolyl (DMIX) group as a superior dummy ligand, which enables not only site-selective C−H functionalization to afford unsymmetrical iodonium salts, but also highly selective aryl transfer during the subsequent metal-free coupling reaction. Both electron-rich and moderately electron-deficient arenes can be converted into the iodonium salts through C−H functionalization, allowing for diverse structural elaboration by metal-free C−N, C−C, C−S, and C−O coupling.
 2022-08-11
2022-08-11
-
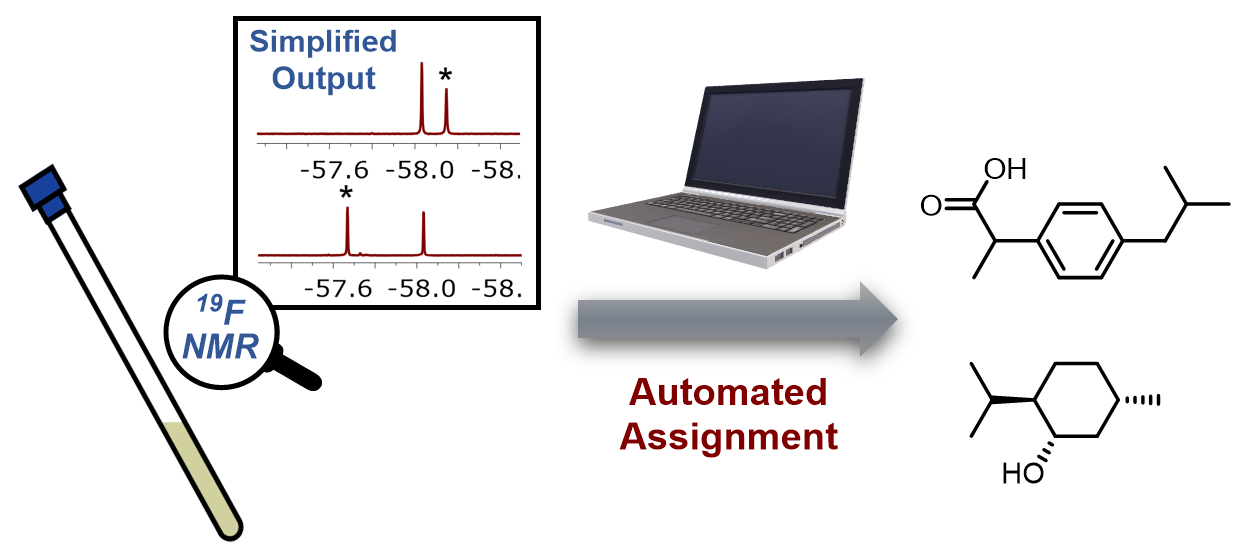
Recognition-Enabled Automated Analyte Identification Via 19F NMR
Nuclear magnetic resonance (NMR) is an indispensable tool for structural elucidation and noninvasive analysis. Automated identification of analytes with NMR is highly pursued in metabolism research and disease diagnosis; however, this process is often complicated by the signal overlap and the sample matrix. We herein report a detection scheme based on 19F NMR spectroscopy and dynamic recognition, which effectively simplifies the detection signal and mitigates the influence of the matrix on the detection. It is demonstrated that this approach can not only detect and differentiate capsaicin and dihydrocapsaicin in complex real-world samples but also quantify the ibuprofen content in sustained-release capsules. Based on the 19F signals obtained in the detection using a set of three 19F probes, automated analyte identification is achieved, effectively reducing the odds of misrecognition caused by structural similarity.
 2022-08-11
2022-08-11
-

021-54925545
-

zhaoyanchuan@sioc.ac.cn
-

Room 1401, Junmou Building, 345 Lingling Road, Xuhui , Shanghai
Copyright © 2020 Yanchuan Zhao Research group of Key Laboratory of Organic Fluorine Chemistry, Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences All rights reserved
沪ICP备09006577号-3




